7. Variant calling¶
7.1. Preface¶
In this section we will use our genome assembly based on the ancestor and call genetic variants in the evolved line [NIELSEN2011].
7.2. Overview¶
The part of the workflow we will work on in this section can be viewed in Fig. 7.1.
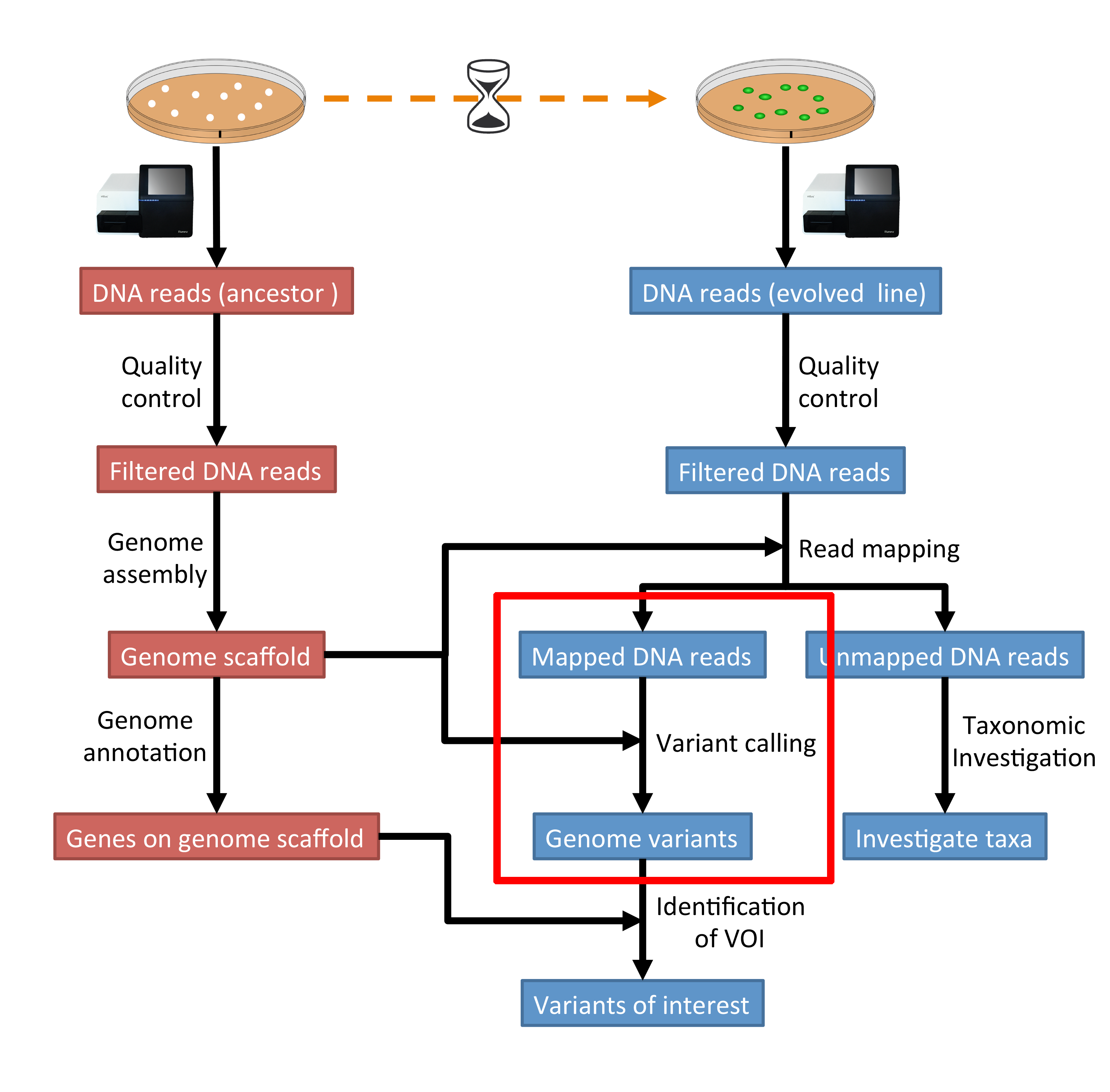
Fig. 7.1 The part of the workflow we will work on in this section marked in red.¶
7.3. Learning outcomes¶
After studying this tutorial section you should be able to:
#. Use tools to call variants based on a reference genome. #, Be able to describe what influences the calling of variants.
7.4. Before we start¶
Lets see how our directory structure looks so far:
cd ~/analysis
ls -1F
assembly/
data/
kraken/
mappings/
trimmed/
trimmed-fastqc/
7.5. Installing necessary software¶
Tools we are going to use in this section and how to intall them if you not have done it yet.
# activate the env
conda activate ngs
# Install these tools into the conda environment
# if not already installed
conda install samtools
conda install bamtools
conda install freebayes
conda install bedtools
conda install vcflib
conda install rtg-tools
conda install bcftools
7.6. Preprocessing¶
We first need to make an index of our reference genome as this is required by the SNP caller.
Given a scaffold/contig file in fasta-format, e.g. scaffolds.fasta which is located in the directory assembly/spades_final, use SAMtools to do this:
samtools faidx assembly/spades_final/scaffolds.fasta
Furthermore we need to pre-process our mapping files a bit further and create a bam-index file (.bai) for the bam-file we want to work with:
bamtools index -in mappings/evolved-6.sorted.dedup.q20.bam
Lets also create a new directory for the variants:
mkdir variants
7.7. Calling variants¶
7.7.1. SAMtools mpileup¶
We use the sorted filtered bam-file that we produced in the mapping step before.
# We first pile up all the reads and then call variants
samtools mpileup -u -g -f assembly/spades_final/scaffolds.fasta mappings/evolved-6.sorted.dedup.q20.bam | bcftools call -v -m -O z -o variants/evolved-6.mpileup.vcf.gz
SAMtools mpileup parameter:
-u: uncompressed output-g: generate genotype likelihoods in BCF format-f FILE: faidx indexed reference sequence file
BCFtools view parameter:
-v: output variant sites only-m: alternative model for multiallelic and rare-variant calling-o: output file-name-O z: output type: ‘z’ compressed VCF
7.7.2. Freebayes¶
As an alternative we can do some variant calling with another tool called freebayes.
Given a reference genome scaffold file in fasta-format, e.g. scaffolds.fasta and the index in .fai format and a mapping file (.bam file) and a mapping index (.bai file), we can call variants with freebayes like so:
# Now we call variants and pipe the results into a new file
freebayes -f assembly/spades_final/scaffolds.fasta mappings/evolved-6.sorted.dedup.q20.bam | gzip > variants/evolved-6.freebayes.vcf.gz
7.8. Post-processing¶
7.8.1. Understanding the output files (.vcf)¶
Lets look at a vcf-file:
# first 10 lines, which are part of the header
zcat variants/evolved-6.mpileup.vcf.gz | head
##fileformat=VCFv4.2
##FILTER=<ID=PASS,Description="All filters passed">
##samtoolsVersion=1.3.1+htslib-1.3.1
##samtoolsCommand=samtools mpileup -g -f assembly/spades_final/scaffolds.fasta -o variants/evolved-6.mpileup.bcf mappings/evolved-6.sorted.q20.bam
##reference=file://assembly/spades_final/scaffolds.fasta
##contig=<ID=NODE_1_length_1419525_cov_15.3898,length=1419525>
##contig=<ID=NODE_2_length_1254443_cov_15.4779,length=1254443>
##contig=<ID=NODE_3_length_972329_cov_15.3966,length=972329>
##contig=<ID=NODE_4_length_951685_cov_15.4231,length=951685>
##contig=<ID=NODE_5_length_925222_cov_15.39,length=925222>
##contig=<ID=NODE_6_length_916533_cov_15.4426,length=916533>
Lets look at the variants:
# remove header lines and look at top 4 entires
zcat variants/evolved-6.mpileup.vcf.gz | egrep -v '##' | head -4
#CHROM POS ID REF ALT QUAL FILTER INFO FORMAT mappings/evolved-6.sorted.q20.bam
NODE_1_length_1419525_cov_15.3898 24721 . T C 164 . DP=12;VDB=0.205941;SGB=-0.680642;MQ0F=0;AC=2;AN=2;DP4=0,0,12,0;MQ=40 GT:PL 1/1:191,36,0
NODE_1_length_1419525_cov_15.3898 157033 . AAGAGAGAGAGAGAGAGAGAGAGA AAGAGAGAGAGAGAGAGAGAGA 39.3328 . INDEL;IDV=6;IMF=0.146341;DP=41;VDB=0.0813946;SGB=-0.616816;MQSB=1;MQ0F=0;ICB=1;HOB=0.5;AC=1;AN=2;DP4=13,17,3,3;MQ=42 GT:PL 0/1:75,0,255
NODE_1_length_1419525_cov_15.3898 162469 . T C 19.609 . DP=16;VDB=0.045681;SGB=-0.511536;RPB=0.032027;MQB=0.832553;BQB=0.130524;MQ0F=0;ICB=1;HOB=0.5;AC=1;AN=2;DP4=13,0,3,0;MQ=39 GT:PL 0/1:54,0,155
The fields in a vcf-file are described in he table (Table 7.1) below:
Col |
Field |
Description |
|---|---|---|
1 |
CHROM |
Chromosome name |
2 |
POS |
1-based position. For an indel, this is the position preceding the indel. |
3 |
ID |
Variant identifier. Usually the dbSNP rsID. |
4 |
REF |
Reference sequence at POS involved in the variant. For a SNP, it is a single base. |
5 |
ALT |
Comma delimited list of alternative seuqence(s). |
6 |
QUAL |
Phred-scaled probability of all samples being homozygous reference. |
7 |
FILTER |
Semicolon delimited list of filters that the variant fails to pass. |
8 |
INFO |
Semicolon delimited list of variant information. |
9 |
FORMAT |
Colon delimited list of the format of individual genotypes in the following fields. |
10+ |
Sample(s) |
Individual genotype information defined by FORMAT. |
7.8.2. Statistics¶
Now we can use it to do some statistics and filter our variant calls.
First, to prepare out vcf-file for querying we need to index it with tabix:
tabix -p vcf variants/evolved-6.mpileup.vcf.gz
-p vcf: input format
We can get some quick stats with rtg vcfstats:
rtg vcfstats variants/evolved-6.mpileup.vcf.gz
Example output from rtg vcfstats:
Location : variants/evolved-6.mpileup.vcf.gz
Failed Filters : 0
Passed Filters : 516
SNPs : 399
MNPs : 0
Insertions : 104
Deletions : 13
Indels : 0
Same as reference : 0
SNP Transitions/Transversions: 1.87 (286/153)
Total Het/Hom ratio : 3.20 (393/123)
SNP Het/Hom ratio : 8.98 (359/40)
MNP Het/Hom ratio : - (0/0)
Insertion Het/Hom ratio : 0.30 (24/80)
Deletion Het/Hom ratio : 3.33 (10/3)
Indel Het/Hom ratio : - (0/0)
Insertion/Deletion ratio : 8.00 (104/13)
Indel/SNP+MNP ratio : 0.29 (117/399)
However, we can also run BCFtools to extract more detailed statistics about our variant calls:
bcftools stats -F assembly/spades_final/scaffolds.fasta -s - variants/evolved-6.mpileup.vcf.gz > variants/evolved-6.mpileup.vcf.gz.stats
-s -: list of samples for sample stats, “-” to include all samples-F FILE: faidx indexed reference sequence file to determine INDEL context
Now we take the stats and make some plots (e.g. Fig. 7.2) which are particular of interest if having multiple samples, as one can easily compare them. However, we are only working with one here:
mkdir variants/plots
plot-vcfstats -p variants/plots/ variants/evolved-6.mpileup.vcf.gz.stats
-p: The output files prefix, add a slash at the end to create a new directory.
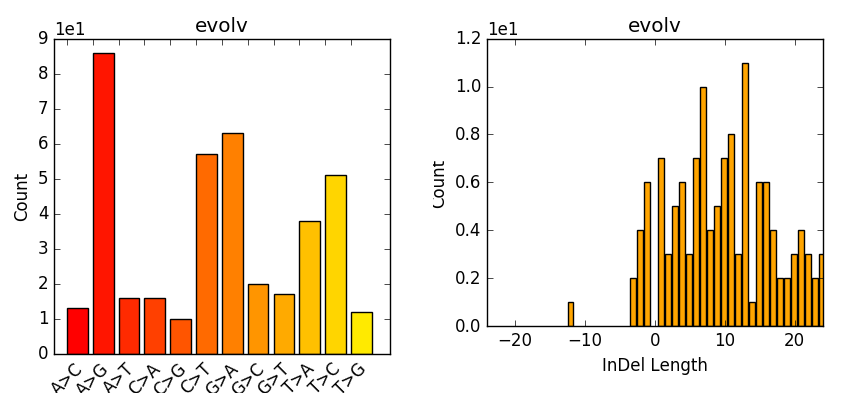
Fig. 7.2 Example of plot-vcfstats output.¶
7.8.3. Variant filtration¶
Variant filtration is a big topic in itself [OLSEN2015]. There is no consens yet and research on how to best filter variants is ongoing.
We will do some simple filtration procedures here. For one, we can filter out low quality reads.
Here, we only include variants that have quality > 30.
# use rtg vcfffilter
rtg vcffilter -q 30 -i variants/evolved-6.mpileup.vcf.gz -o variants/evolved-6.mpileup.q30.vcf.gz
-i FILE: input file-o FILE: output file-q FLOAT: minimal allowed quality in output.
or use vcflib:
# or use vcflib
zcat variants/evolved-6.mpileup.vcf.gz | vcffilter -f "QUAL >= 30" | gzip > variants/evolved-6.mpileup.q30.vcf.gz z
-f "QUAL >= 30": we only include variants that have been called with quality >= 30.
Quick stats for the filtered variants:
# look at stats for filtered
rtg vcfstats variants/evolved-6.mpileup.q30.vcf.gz
freebayes adds some extra information to the vcf-files it creates. This allows for some more detailed filtering. This strategy will NOT work on the SAMtools mpileup called variants Here we filter, based on some recommendation form the developer of freebayes:
zcat variants/evolved-6.freebayes.vcf.gz | vcffilter -f "QUAL > 1 & QUAL / AO > 10 & SAF > 0 & SAR > 0 & RPR > 1 & RPL > 1" | gzip > variants/evolved-6.freebayes.filtered.vcf.gz
QUAL > 1: removes really bad sitesQUAL / AO > 10: additional contribution of each obs should be 10 log units (~ Q10 per read)SAF > 0 & SAR > 0: reads on both strandsRPR > 1 & RPL > 1: at least two reads “balanced” to each side of the site
Todo
Look at the statistics. One ratio that is mentioned in the statistics is transition transversion ratio (ts/tv). Explain what this ratio is and why the observed ratio makes sense.
This strategy used here will do for our purposes. However, several more elaborate filtering strategies have been explored, e.g. here.
References
- NIELSEN2011
Nielsen R, Paul JS, Albrechtsen A, Song YS. Genotype and SNP calling from next-generation sequencing data. Nat Rev Genetics, 2011, 12:433-451
- OLSEN2015
Olsen ND et al. Best practices for evaluating single nucleotide variant calling methods for microbial genomics. Front. Genet., 2015, 6:235.